A unique drug, NVG-111, first discovered and then developed by a research team from UCL.
Writer: Maja Bronowska
Editor: Chelsea Tripp
Artist: Lucie Gourmet
Cancer, often called an emperor of all maladies, affected 18.1 million people worldwide in 2018 alone. The incidence of the disease, unfortunately, continues to create a huge burden to society. The need for new treatments is urgent; however, the pipeline of novel drug discoveries is extremely challenging. Clinical trials are a significant part of that process. In June 2021, the National Institute for Health Research at University College London Hospital (UCLH) Clinical Research Facility announced a new clinical trial for Non-Hodgkin Lymphomas (NHL) subtypes, in which they will investigate an innovative drug – NVG-111.
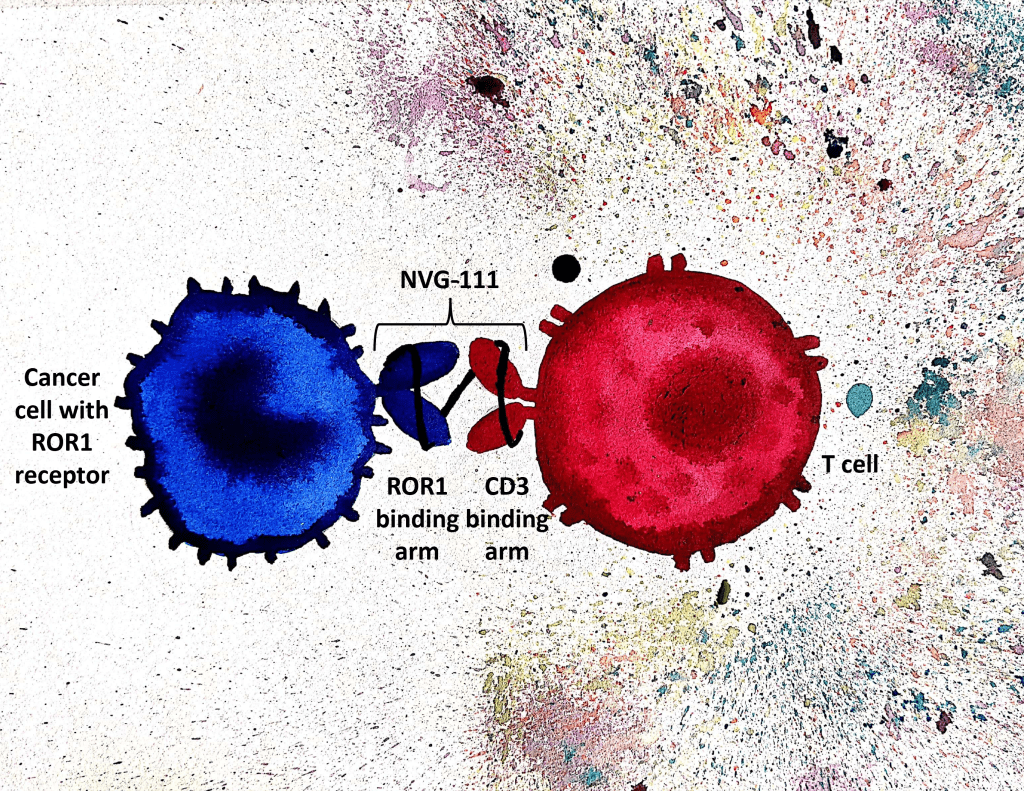
The drug was developed by researchers from UCL Immunity & Transplantation led by Professor Amit Nathwani. NVG-111, a monoclonal antibody, also known as NovalGen, is the first such drug that targets a tyrosine kinase receptor, ROR1, which is an antigen present on various malignancies and cancer-initiating cells. The uniqueness of the drug is that it is a bi-specific T cell engager. In other words, NVG-111 works as a bridge between cancer cells and cytotoxic T cells. The antibody binds two antigens – one specific for the neoplastic cell and the other present on the T cell surface.
T cells together with B cells are a part of the adaptive immunity, which is highly specific to foreign pathogens. The main challenge in targeting cancer is that it develops from our own cells. The immune system is only capable of recognising damaged cells or non-self antigens (pathogens). Thus, the mutated cells often remain unrecognised by the immune system and survive. Neoplasms outwit the immune system and take advantage of the mechanisms used by healthy cells. The main role of B cells is to produce antibodies whereas T cells use different mechanisms to recognise damaged (in this case mutated cells) or non-self cells and alert the whole immune system. NVG-111 activates T cell engagers which stimulate the immune system without depleting the B cell pool in the organism.
The drug has the potential to target selected cancer stem cells, which often escape from the standard treatment and drive cancer evolutionary processes, making the diseases resistant to the treatment and increasing the risk of recurrence. The novelty of NVG-111 allows optimal activation of T-cells and avoids the excessive release of cytokines (small proteins regulating the intensity and directing immune responses), which is often harmful to healthy tissues. Thus, NVG-11 prompts the immune cells to only kill the cancerous ones.
Based on preclinical studies, not only is the drug believed to be effective for solid tumours (e.g., pancreatic cancer), but also for haematological malignancies (e.g., lymphoma). Additionally, the treatment enters the most innovative field of immunotherapies, alongside therapies such as chimeric antigen receptor (CAR) T-cell or T cell engineered receptor (TCR) cell therapies. However, the mechanisms of killing the cancer cells by stimulating the receptor are not dependent on the specificity of the immune system in terms of major histocompatibility complex (MHC) nor antigen presentation. MHC is a class of molecules that is specific to the individual and used by the immune system to assess if the cell is self or non-self.
NVG-111 does not require ex vivo engineering nor manipulation of T cells – making the treatment more standardised for all patients and cost-effective. The drug consists of two small fragments of antibodies, where one targets T cells and the other one to the surface antigen characteristic for cancer. In this case, it is ROR1. This therapy is promising; however, a first-in-human trial is crucial to assess the efficacy and safety of the treatment.
The trial will be led by multiple centres in the UK, including the UCLH hospital. The drug has the potential to work for solid tumours, but, firstly, it will be trialled on patients suffering from NHLs such as lymphocytic leukaemia, small lymphocytic lymphoma and mantle cell lymphoma. Currently, those subtypes are very resistant to treatment and often are thought to be incurable. Thus, the trial brings hope for many people and the future of oncological patients’ care.
As the team reports, the trial will be divided into two parts, A and B. The main goal of part A will be to establish adequate dosing based on “dose-limiting toxicities”. To do so, sequential doses will be given to the patients and the side effects will be evaluated. Part B will be challenged with assessing the further efficacy of the treatment and finding the NVG-111 safety profile before engaging in further stages of the trial.
Cancer treatment still has an enormous demand for new therapies. Many patients are left hopeless after the diagnosis with a definitive cure still out of reach. This trial may change that and bring the prospect of new therapies being created by UCL applying translational medicine into real solutions.
